Minnesota CWD investigation spurred by Douglas County deer farm detection concludes
News Release
For immediate release: March 13, 2020
Contact: Erin Crider
Investigation confirms four additional CWD positive deer on Pine County farm
Test results from the depopulation of a Pine County deer farm have confirmed four additional cases chronic wasting disease (CWD). The first CWD positive animals at this farm were confirmed in January 2020, resulting in depopulation of the herd. This herd was investigated because it provided animals to a Douglas County deer farm in the past, including a CWD-positive doe that began the disease investigation in December 2019.
“This CWD investigation has gone very well,” said Board Assistant Director, Dr. Linda Glaser. “We identified the Pine County herd quickly from that initial positive result in Douglas County. We immediately traced animals and quarantined herds. Those actions were in coordination with other agencies to rapidly respond to CWD in Minnesota.”
In early December 2019, a doe from the Pine County herd died, was tested and found to be CWD positive. In January, two fawns were harvested, and one tested positive for CWD. Following this development, the remaining six deer in the herd were depopulated, all of which were submitted for CWD testing. Results from the National Veterinary Services Laboratory on these six deer confirmed the detection of CWD in two does and one fawn. In all, five of the nine total animals in the Pine County herd were CWD positive.
The Pine County and Douglas County sites are not allowed to have any deer or elk for five years. Owners must maintain fencing to prevent wild deer from accessing empty pens. Biohazard signs have been posted on the fencing and must be maintained for the entire five-year fallow period. The investigation is ongoing, and the Board will continue to take immediate action if any new detections are identified.
CWD is a disease of the deer and elk family caused by prions, which can damage brain and nerve tissue. The disease is most likely transmitted when infected deer and elk shed prions in saliva, feces, urine, and other fluids or tissues. CWD is not known to naturally occur in other animals. The disease is fatal in deer and elk, and there are no known treatments or vaccines. Consuming infected meat is not advised.
--30--
News Release
For immediate release: January 10, 2020
Contact: Michael Crusan
Investigation leads to additional CWD positive deer on Pine County farm
News Release
Wild deer in Dakota County confirmed positive for chronic wasting disease
March 13, 2020
Discovery marks first case of CWD in this area of Minnesota
A wild deer in Dakota County was confirmed positive for chronic wasting disease, the Minnesota Department of Natural Resources said.
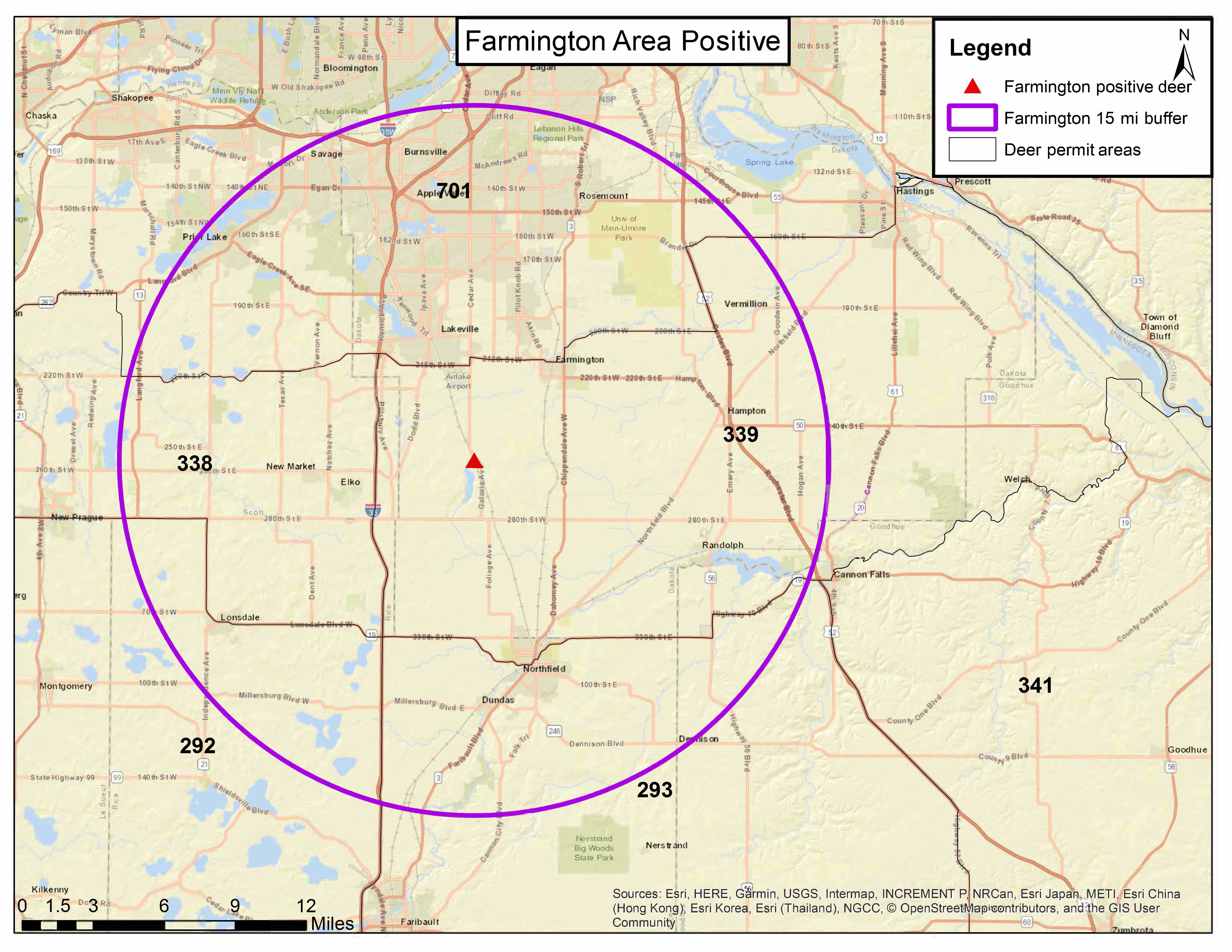
The deer, an adult male, was reported by a local resident near Farmington as displaying neurological symptoms and was tested as part of the DNR’s risk-based disease surveillance program.
It is the first detection of the fatal neurological disease in a wild deer in this county, and this deer was found nearly 100 miles from the state’s primary CWD area near Preston, Minnesota.
“An informed citizen did the right thing by calling DNR, which allowed us to identify and remove this deer from the landscape,” said Lou Cornicelli, DNR wildlife research manager. “We’re hopeful the disease is not widespread in the area.”
In the short term, the DNR is developing plans to sample deer opportunistically until the fall hunting season. Cornicelli said deer hunting is the primary tool for managing this disease and the DNR will follow its CWD response plan PDFto identify a CWD management zone that will be at least 15 miles around the positive deer.
Hunters can expect to see carcass movement restrictions and mandatory surveillance. People who are unfamiliar with how deer are managed in Minnesota can find deer-related information, including hunting, natural history of deer and the state’s deer management plan, on the DNR deer management webpage. The DNR will work closely with tribal communities and with cities, townships and counties to manage this disease collaboratively.
In addition, the DNR will prohibit recreational deer feeding. Until then, the DNR asks that residents voluntarily stop feeding deer.
The Board of Animal Health, which oversees farmed deer and elk in the state, is expanding its endemic area for CWD based on this new detection by the DNR. The Board establishes the endemic area boundary 15 miles around all confirmed cases of CWD in the wild.
Additional CWD information
CWD affects the cervid family, which includes deer, elk and moose. The disease is not known to affect human or pet health. It is spread through direct contact with an infected deer’s saliva, urine, blood, feces, antler velvet or carcass. There is no vaccine or treatment for this disease.
For more information on chronic wasting disease, including maps of CWD surveillance areas, frequently asked questions and hunter information, visit mndnr.gov/cwd.
South metro CWD
Discovery
 A resident near Farmington contacted DNR to report an adult male deer on their property that was displaying neurological symptoms such as being unafraid of humans, stumbling, swaying and moving with its head hanging low.
A resident near Farmington contacted DNR to report an adult male deer on their property that was displaying neurological symptoms such as being unafraid of humans, stumbling, swaying and moving with its head hanging low.Deer feeding bans
Next steps
- The DNR is currently developing plans to sample deer opportunistically until the fall hunting season.
- The DNR will work to identify a CWD management zone that will be at least 15 miles around the positive deer, in accordance to its CWD response plan.
- This fall, hunters can expect to see carcass movement restrictions and mandatory surveillance.
- People who are unfamiliar with deer management are encouraged to spend some time on the DNR's deer management page.
i think some hunters that don't read this carefully are going to think this is a cure all for cwd tse contamination. IT'S NOT!
first off, it would take a strong bleach type sodium hypochlorite, that is NOT your moms bleach she uses in her clothes, and store bought stuff.
Concentrated bleach is an 8.25 percent solution of sodium hypochlorite, up from the “regular bleach” concentration of 5.25 percent.Nov 1, 2013 https://waterandhealth.org/disinfect/high-strength-bleach-2/
second off, the study states plainly;
''We found that a five-minute treatment with a 40% dilution of household bleach was effective at inactivating CWD seeding activity from stainless-steel wires and CWD-infected brain homogenates. However, bleach was not able to inactivate CWD seeding activity from solid tissues in our studies.''
''We initially tested brains from two CWD-infected mice and one uninfected mouse using 40% bleach for 5 minutes. The results from these experiments showed almost no elimination of prion seeding activity (Table 4). We then increased the treatment time to 30 minutes and tested 40% and 100% bleach treatments. Again, the results were disappointing and showed less than a 10-fold decrease in CWD-seeding activity (Table 4). Clearly, bleach is not able to inactivate prions effectively from small brain pieces under the conditions tested here.''
''We found that both the concentration of bleach and the time of treatment are critical for inactivation of CWD prions. A 40% bleach treatment for 5 minutes successfully eliminated detectable prion seeding activity from both CWD-positive brain homogenate and stainless-steel wires bound with CWD. However, even small solid pieces of CWD-infected brain were not successfully decontaminated with the use of bleach.''
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0223659
https://chronic-wasting-disease.blogspot.com/2019/10/inactivation-of-chronic-wasting-disease.html
i think with all the fear from recent studies, and there are many, of potential, or likelihood of zoonosis, if it has not already happened as scjd, i think this study came out to help out on some of that fear, that maybe something will help, but the study plainly states it's for sure not a cure all for exposure and contamination of the cwd tse prion on surface materials. imo...terry
172. Establishment of PrPCWD extraction and detection methods in the farm soil
186. Serial detection of hematogenous prions in CWD-infected deer
Our ongoing studies will show whether the transmission of CWD into macaques and passage in transgenic mice represents a form of non-adaptive prion amplification, and whether macaque-adapted prions have the potential to infect mice expressing human PrP.
states.
https://www.nature.com/articles/srep11573
Prion diseases (PD) are the unique neurodegenerative proteinopathies reputed to be transmissible under field conditions since decades. The transmission of Bovine Spongiform Encephalopathy (BSE) to humans evidenced that an animal PD might be zoonotic under appropriate conditions. Contrarily, in the absence of obvious (epidemiological or experimental) elements supporting a transmission or genetic predispositions, PD, like the other proteinopathies, are reputed to occur spontaneously (atpical animal prion strains, sporadic CJD summing 80% of human prion cases).
Non-human primate models provided the first evidences supporting the transmissibiity of human prion strains and the zoonotic potential of BSE. Among them, cynomolgus macaques brought major information for BSE risk assessment for human health (Chen, 2014), according to their phylogenetic proximity to humans and extended lifetime. We used this model to assess the zoonotic potential of other animal PD from bovine, ovine and cervid origins even after very long silent incubation periods.
*** We recently observed the direct transmission of a natural classical scrapie isolate to macaque after a 10-year silent incubation period,
***with features similar to some reported for human cases of sporadic CJD, albeit requiring fourfold long incubation than BSE. Scrapie, as recently evoked in humanized mice (Cassard, 2014),
***is the third potentially zoonotic PD (with BSE and L-type BSE),
***thus questioning the origin of human sporadic cases.
We will present an updated panorama of our different transmission studies and discuss the implications of such extended incubation periods on risk assessment of animal PD for human health.
===============
***thus questioning the origin of human sporadic cases***
===============
***our findings suggest that possible transmission risk of H-type BSE to sheep and human. Bioassay will be required to determine whether the PMCA products are infectious to these animals.
==============
https://prion2015.files.wordpress.com/2015/05/prion2015abstracts.pdf
***Transmission data also revealed that several scrapie prions propagate in HuPrP-Tg mice with efficiency comparable to that of cattle BSE. While the efficiency of transmission at primary passage was low, subsequent passages resulted in a highly virulent prion disease in both Met129 and Val129 mice.
***Transmission of the different scrapie isolates in these mice leads to the emergence of prion strain phenotypes that showed similar characteristics to those displayed by MM1 or VV2 sCJD prion.
***These results demonstrate that scrapie prions have a zoonotic potential and raise new questions about the possible link between animal and human prions.
http://www.tandfonline.com/doi/abs/10.1080/19336896.2016.1163048?journalCode=kprn20
PRION 2016 TOKYO
Saturday, April 23, 2016
SCRAPIE WS-01: Prion diseases in animals and zoonotic potential 2016
Prion. 10:S15-S21. 2016 ISSN: 1933-6896 printl 1933-690X online
Taylor & Francis
Prion 2016 Animal Prion Disease Workshop Abstracts
WS-01: Prion diseases in animals and zoonotic potential
Transmission of the different scrapie isolates in these mice leads to the emergence of prion strain phenotypes that showed similar characteristics to those displayed by MM1 or VV2 sCJD prion.
These results demonstrate that scrapie prions have a zoonotic potential and raise new questions about the possible link between animal and human prions.
http://www.tandfonline.com/doi/abs/10.1080/19336896.2016.1163048?journalCode=kprn20
Title: Transmission of scrapie prions to primate after an extended silent incubation period)
*** In complement to the recent demonstration that humanized mice are susceptible to scrapie, we report here the first observation of direct transmission of a natural classical scrapie isolate to a macaque after a 10-year incubation period. Neuropathologic examination revealed all of the features of a prion disease: spongiform change, neuronal loss, and accumulation of PrPres throughout the CNS.
*** This observation strengthens the questioning of the harmlessness of scrapie to humans, at a time when protective measures for human and animal health are being dismantled and reduced as c-BSE is considered controlled and being eradicated.
*** Our results underscore the importance of precautionary and protective measures and the necessity for long-term experimental transmission studies to assess the zoonotic potential of other animal prion strains.
http://www.ars.usda.gov/research/publications/publications.htm?SEQ_NO_115=313160
